The kinetic theory of gases allows deducing the properties of the ideal gas using a model in which the gas molecules are spheres that comply with the laws of classical mechanics.
The properties that can be calculated using this model are: gas pressure, molecular velocity distribution, mean molecular velocity, collision velocity, and mean distance between collisions. These properties allow the study of the kinetics of reactions in the gas phase as well as the flow of fluids and heat transfer.
Fundamentals of the kinetic-molecular theory of gases
 Kinetic theory can be considered as a branch of statistical thermodynamics since it deduces macroscopic properties of matter from molecular properties. The principles on which it is based are the following:
Kinetic theory can be considered as a branch of statistical thermodynamics since it deduces macroscopic properties of matter from molecular properties. The principles on which it is based are the following:
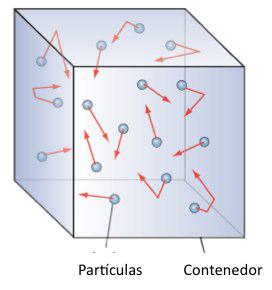
- A gas is made up of a large number of spherical particles whose size is negligible compared to the distance between the particles.
- Molecules move in straight lines at high speed and only interact when they collide. The collisions between particles and with the walls of the container are considered perfectly elastic, conserving the translational kinetic energy.
- Kinetic theory assumes that particles obey Newton's laws. This assumption is incorrect (molecules obey the laws of quantum mechanics) and leads to incorrect results in predicting the heat capacities of the gas, although it gives acceptable results in properties such as pressure or diffusion.



